Seaweed Metabolites for Targeting Pel Polysaccharide Biosynthesis in Pseudomonas aeruginosa– A Novel Strategy for Biofilm Control
DOI:
https://doi.org/10.5530/ctbp.2023.3s.66Keywords:
Seaweeds, Sargassum sp, Pseudomonas aeruginosa, Biofilm, Inhibitor, Pel DAbstract
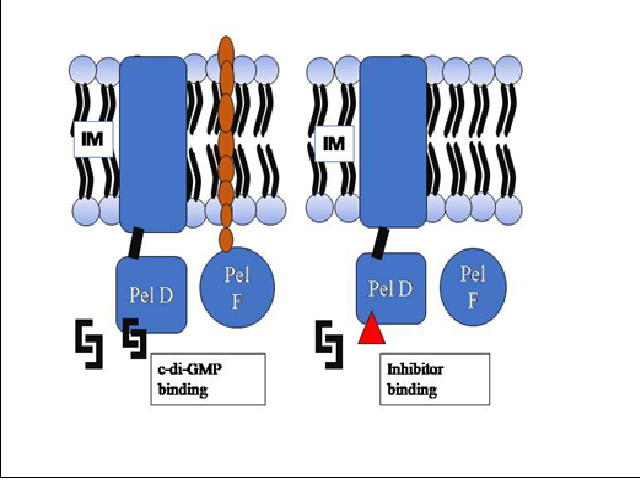
According to the National Institute of Health, more than 80% of microbial infections are biofilm-associated, including chronic lung, wound and ear infections. Pseudomonas aeruginosa is an opportunistic pathogen that causes chronic nosocomial infection in immune-compromised and cystic fibrosis patients worldwide and is a common cause of urinary tract and burn infections. Its ability to form biofilm makes them highly resistant to antibiotics and the host’s immune defense system. Biofilm produced by Pseudomonas aeruginosa is made up of exopolysaccharides such as pel, alginate and psl. Different strategies are reported in the control of biofilm formation. The present work focuses on targeting pellicle (pel) polysaccharide biosynthesis in Pseudomonas aeruginosa using a computational approach. Pel polysaccharide biosynthesis involves seven proteins present in the cytoplasmic, inner, periplasmic and outer membranes of Pseudomonas aeruginosa. In the current study, Pel D was taken as the target for inhibition of pel polysaccharide production. The marine environment provides a novel array of metabolites with unique structural features and bioactivities. Recently, metabolites derived from seaweeds or marine macroalgae have attracted increasing consideration for the development of novel drugs. In this perspective, thirty-nine metabolites reported in brown seaweed Sargassum were chosen as the source for druggable molecules (ligands). The structures of these metabolites were retrieved from the Seaweed Metabolite Database. Molecular docking was performed using PyRx software. Four metabolites of Sargassum were found to exhibit high binding affinity with target protein when compared to ibuprofen, a non-steroidal anti-inflammatory candidate drug reported for inhibition of biofilm control in Pseudomonas aeruginosa. The top four metabolites with high binding affinity were assessed for physicochemical, pharmacokinetic and drug-likeness properties through in silico analysis. Hence this study will pave the direction for exploring the potential of less explored seaweed metabolites as therapeutic mediators for the control of Pseudomonas biofilm infection.