A Novel Sustained-Release In-Situ Implant of Fluvoxamine Ameliorates Depressive-Like Behaviours in a CUMS Mouse Model
DOI:
https://doi.org/10.5530/ctbp.2026.2.14Keywords:
Fluvoxamine, Sustained-release implant, In situ forming system, Depression, Mental health, SocioeconomicAbstract
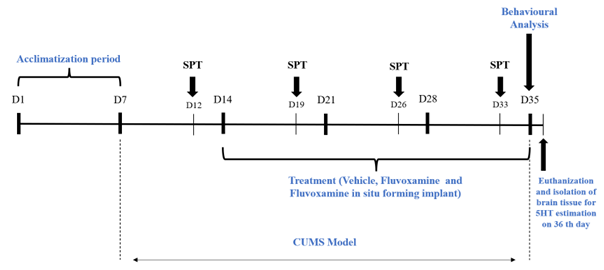
Nearly 280 million individuals worldwide suffer from depression, a serious mental health condition. Numerous antidepressants are available; however, treatment results are still not ideal because of factors like low adherence, frequent dosage schedules, delayed start, and uneven efficacy among patient populations. Because it blocks the serotonin transporter (SERT) and modifies sigma-1 receptor activation, fluvoxamine, a selective serotonin reuptake inhibitor (SSRI), is used extensively. However, pharmacokinetic restrictions that need two or three daily dosages frequently limit its therapeutic potential. This makes a strong case for the creation of innovative drug delivery methods that guarantee continuous drug release, preserve steady plasma levels, and eventually enhance longterm effectiveness and patient adherence. To get around the drawbacks of traditional dosage, the current study set out to create and assess a fluvoxamine in situ-forming implant (FISFI) as a sustained-release delivery method. In order to deliver fluvoxamine with a prolonged release in a chronic unpredictable mild stress (CUMS) mouse model of depression, the implant was created using biodegradable polymers. According to the study, both therapy groups showed notable improvements in both biochemical and behavioral indicators. With the lowest immobility durations (63.21 ± 17.95 s), the strongest sucrose preference at day 33, and the lowest serotonin restoration (~648 pg/mg), the FISFI group demonstrated similar efficacy. Individual results for the implant group were more consistent, indicating less variability. These findings demonstrate that fluvoxamine uses sigma-1 receptor activation and serotonergic modulation to produce strong antidepressant-like effects. By incorporating fluvoxamine into a biodegradable polymeric depot, FISFI may improve clinical results by lowering patient burden and delivering reliable therapeutic effects.